The Many Uses of Salt from the Earth
by Susan Ward Aber
Emporia State University, Emporia, Kansas USA
Introduction
Halite has been historically important and continues to be a resource of major importance for society today. While the price quoted by a Kansas producer was $12.00 per ton for rock salt (Lee Spence, personal communication, October 28, 2005), Hansen (1995) wrote that in the late 1700s and early 1800s salt was "brought by packhorse across the Appalachian Mountains and commanded a price of $4 to $6 dollars per bushel." The United States is the largest salt producer in the world and it is mined in every U.S. state (Klein, 2002, p. 399). The major halite producing states are New York, Ohio, Kansas, Louisiana, Texas, and California (Gwynn, 1996, p. 16). This webpage was designed to provide a window into this fascinating mineral. In addition to a brief introduction to the formation of this mineral and its resulting properties, some of the uses of halite and rock salt will be covered in text and images.
Properties
Halite is a mineral in the halide chemical classification with a composition of NaCl or sodium chloride with ionic bonding. Halite is in the isometric crystal system and as such, displays the highest symmetry in the mineral kingdom. It exhibits the 4/m Bar3 2/m point group and is often found in a hexahedral or cube crystal form. This crystal system means the sodium chloride ions are arranged in a configuration with three crystallographic axes equal in length that meet at 90 degree angles. Furthermore, halite has characteristic three-directional cleavage at right angles, so that when it is subjected to external stress it will break along these planes of atomic weakness and again, result in a cubic shape. Physical properties include a hardness of 2.5 and specific gravity of 2.16. The habit is commonly equant or drusy, while the luster may be waxy to vitreous depending on whether it is translucent or transparent. Color varies with impurities and can be white, yellow, red, blue and purple, as well as colorless when it is in a pure state.
Genesis
Halite is an evaporite mineral found in sedimentary rock environments. It is derived from salt lakes found in enclosed basins, as well as oceans and salt springs. When the water evaporates, the salt is left to precipitate on the Earth's surface. Subsequent sediments cover the salt, and the weight and pressure compact the mineral into solid rock layers.
The Hutchinson Salt Member of the Permian Wellington Formation is an example of an evaporite deposit. The extent of this bedded salt deposit is 27,000 square miles in central and south-central Kansas (Walters, 1978, p. iii). The Hutchinson Salt is marginal to the Permian Basin salt deposits of Oklahoma, Texas Panhandle, and southeastern New Mexico that covers 100,000 square miles (p. 3). See Figure 1 and 2 for geographic and stratigraphic positioning of the salt layer.
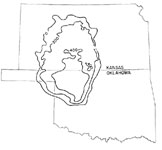
Figure 1. The extent and thickness of
the Hutchinson Salt Member of
the Permian Wellington Formation.
Image scanned from Walters,
1978, p. 3. Also found at the
Kansas Geologic Survey website
www.kgs.ku.edu/Hydro/
Hutch/GeneralGeology.
|
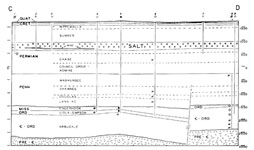
Figure 2. Stratigraphic cross-section of
rock layers near Lyons, Kansas.
Scanned from Walters, 1978, p. 6.
Also see the Kansas Geologic
Survey website
www.kgs.ku.edu/Hydro/
Hutch/GeneralGeology/.
|
Near Hutchinson, The Salt City, the bedded salt layer is 350 feet thick and interbedded with nearly 20% shale, anhydrite, and dolomite (Walters, 1978, p. 13). Because the depth of the salt varies from 200 feet to over 2500 feet below the surface, some of the salt is naturally dissolved by present-day water tables in the eastern extent of the outcrop; however, in the western extent of the outcrop, no instances of dissolution in the Hutchinson Salt occurred until after drilling took place for oil (p. iii). Drilling inadvertently created dissolution of the salt and subsequent subsidence of the surface. For example, in 1914 subsidence associated with the salt works of the Joy Morton Salt Company caused a 150 foot horizontal depression with a vertical drop of 15 feet (p. iii). An example of extreme subsidence related to oil drilling happened in 1959 near Ellinwood, Kansas when "a circular pit 300 feet in diameter and 85 feet deep developed in a few hours around a plugged and abandoned salt water disposal well 3850 feet deep in which the Hutchinson Salt was penetrated from 975 feet to 1275 feet" (p. iii).
 |
Figure 3. A stabilized 300 foot diameter sinkhole, with 18 feet water depth, in southeast Hutchinson, Kansas. The subsidence began a month before this photograph was taken and the sinkhole was associated with the Cargill salt processing company. The railroad tracks had to be relocated. This image was scanned from Walters, 1978, p. 21; originally it was printed in The Wichita Eagle and Beacon newspaper on November 12, 1974. See the Kansas Geologic Survey website for more images:
www.kgs.ku.edu/Hydro/Hutch/Subsidence/.
|
|
Figure 4. This sinkhole developed around an oil well in Barton County, Kansas. The rapid subsidence was cone-shaped and filled with water within hours. The vertical water well casing can be seen as straight line on the left center portion of the sinkhole and a person is standing outside the sinkhole toward the lower left of the picture. This image was scanned from Walters, 1978, p. 55; the photo was taken by Larry Panning, April 24, 1959. See the Kansas Geologic Survey website for schematic drawings of a sinkhole:
www.kgs.ku.edu/Hydro/Hutch/Subsidence/.
|
 |
Uses
Halite, commonly known as salt, has many uses. In fact, a Carey Salt brochure states, "it is estimated that there are over 14,000 different uses for salt" (Carey Salt, n.d.). Halite is a seasoning and preservative for food, although one of the greatest use is for the manufacture of hydrochloric acid (Klein, 2002, p. 400). When salt is found at the Earth's surface, it can be harvested for products or when left untouched, it can create important wetland environments. When salt is found underground, it can be mined for products with the resulting cavities providing efficient storage or museum facilities. If fresh water is pumped into a salt layer, dissolution will occur, which creates underground storage areas for product storage. In other applications of dissolution, deep wells are used to inject fresh water and thus, dissolve the salt and produce a brine that is brought to the surface and boiled in large closed kettles to evaporate pure salt crystals. After screening for the proper size, this salt is boxed and ready for many purposes including food seasoning.
Rock Salt Mining and Storage Caverns
Salt Museums and Environments
References
- Carey Salt (n.d.). Salt: The most precious mineral in the world. 1800 Carey Boulevard, Hutchinson, KS 67501 [Brochure.]
- Gwynn, J. W. (1996). Commonly asked questions about Utah's Great Salt Lake and Ancient Lake Bonneville. Utah Geological Survey, Public Information Series 39, pp. 22.
- Hansen, M. C. (1995). The scioto saline-Ohio's early salt industry. Ohio Department of Natural Resources, Division of Geological Survey, GeoFacts No. 7.
- Kansas Geological Survey (2001-2005). Hutchinson response project: Geology. WWW URL
www.kgs.ku.edu/Hydro/Hutch/GeneralGeology/. Retrieved December 8, 2005.
- Kansas Geological Survey (2001-2005). Hutchinson response project: Land subsidence. WWW URL
www.kgs.ku.edu/Hydro/Hutch/Subsidence/. Retrieved December 8, 2005.
- Klein, C. (2002). The 22nd edition of the manual of mineral science. NY: John Wiley and Sons, Inc.
- Walters, R. F. (1978). Land subsidence in central Kansas related to salt dissolution. Kansas Geological Survey Bulletin 214.
© Notice: The Many Uses of Salt from the Earth webpage was created for educational purposes and while everyone is welcome to view these webpages, any other use or repackaging of these materials is prohibited without permission from the author, Dr. Susan W. Aber, abersusa@emporia.edu. Dr. Aber teaches at Emporia State University, www.emporia.edu/earthsci/abersusa.htm. This webpage was created November 28, 2005; last updated May 30, 2007.



